
Nonetheless, the only way to get the "correct answer" is to do a proper Born-Haber analysis of the data provided. View Notes - Lattice Energy Problems from CHM 115 at Purdue. In most cases, the change is less than 1%.
Most of the time, the missing heat will be the lattice energy, but, occasionally, it will be another term. complete reaction and no other source of magnesium.

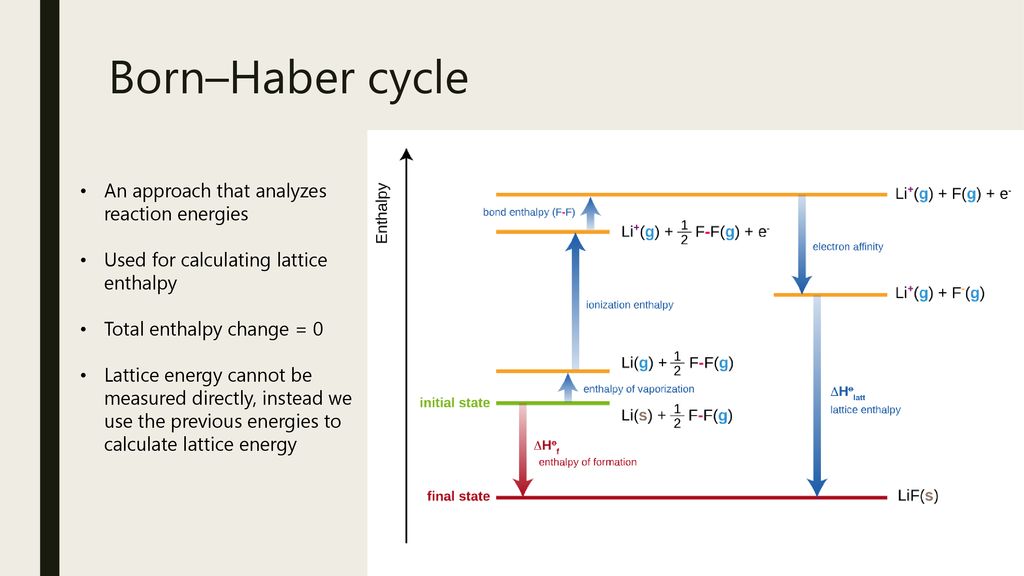
When you press "New Problem", a window will open with a set of thermochemical data related to an ionic compound. The first ionization energies, in kJ mol-1, of four elements with. You must write all thermochemical equations for the steps of the cycle. This page provides exercises in the use of the Born-Haber cycle to determine lattice energies of ionic compounds. Draw Born-Haber cycle for the formation of strontium chloride b.Use the following data to calculate the enthalpy of formation of strontium chloride.
